Peptide synthesis: core methods, applications, innovations

TL;DR:
- Peptide synthesis enables precise creation of therapeutic peptides not obtainable by natural extraction.
- Solid-phase and solution-phase are the main methods, each suited for specific sequence lengths and scale.
- Green chemistry advances aim to reduce toxic solvents and waste, making peptide production safer and more sustainable.
Most therapeutic peptides used in modern medicine cannot be harvested from living organisms in clinically meaningful quantities. Semaglutide, for instance, is a GLP-1 receptor agonist that exists only because precise artificial chemical synthesis makes large-scale production possible. This reality places peptide synthesis at the absolute center of drug development and biotechnology. Understanding the methods, the chemistry, and the emerging innovations behind this process is no longer optional for researchers and professionals working in therapeutics. This guide covers the core synthesis strategies, the workflow challenges you will actually encounter in the lab, and the green chemistry trends reshaping the field.
Table of Contents
- What is peptide synthesis and why does it matter?
- Core methods: solid-phase vs. solution-phase peptide synthesis
- Critical steps and challenges in the synthesis process
- Green chemistry and the future of peptide synthesis
- What most professionals miss about peptide synthesis
- Explore advanced peptide solutions and resources
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Essential drug development tool | Peptide synthesis enables creation of custom peptides critical to modern therapeutics. |
| Method choice impacts outcomes | Selecting between SPPS and solution-phase methods affects efficiency, scale, and purity. |
| Workflow optimization reduces risks | Addressing side reactions and using proper protections improves peptide yield and safety. |
| Sustainable practices are advancing | Green chemistry and AI are making peptide synthesis safer and more efficient for the future. |
What is peptide synthesis and why does it matter?
At its core, peptide synthesis is the chemical process of forming peptide bonds between amino acids to create defined peptide sequences. Unlike proteins expressed by living cells, synthetic peptides are built one residue at a time under controlled laboratory conditions, giving researchers exact control over sequence, length, and modification. That level of precision is simply not achievable through biological extraction.
Naturally occurring peptides can be isolated from tissues or fermentation broths, but yields are low, purification is costly, and sequence modification is impossible after the fact. Synthetic routes eliminate those constraints. You can introduce non-natural amino acids, add pegylation sites, incorporate isotopic labels for imaging studies, or attach cytotoxic payloads for antibody-drug conjugates. The flexibility is the point.
“Peptide synthesis is foundational to producing therapeutic peptides used in medicine, enabling the precise manufacture of sequences that cannot be reliably obtained from biological sources.”
The pharmaceutical relevance is enormous. GLP-1 agonists like semaglutide and tirzepatide, oxytocin analogs, antimicrobial peptides, and cancer immunotherapy epitopes all depend on synthetic production. Beyond therapeutics, synthetic peptides power proteomics research, enzyme inhibition studies, receptor binding assays, and vaccine development.
Typical uses of synthetic peptides in research and medicine:
- Development of GLP-1 agonists and other metabolic disease drugs
- Epitope mapping and vaccine antigen production
- Protease substrate libraries for drug target validation
- Radiolabeled peptides for diagnostic imaging
- Antimicrobial peptide screening programs
- Biomarker discovery and mass spectrometry calibration standards
- Peptide-drug conjugates for targeted oncology therapies
For a broader look at how synthetic peptides fit into current research priorities, the science-based peptide guide at Primegen Labs covers the biological mechanisms in depth. Regulatory context is equally important, and peptide regulations in 2026 have shifted significantly for researchers working toward clinical translation.
Core methods: solid-phase vs. solution-phase peptide synthesis
Two primary strategies dominate the field: solid-phase peptide synthesis (SPPS) and liquid-phase peptide synthesis (LPPS, also called solution-phase). Each has a distinct workflow logic, and choosing between them is not just a matter of tradition.
SPPS is the dominant methodology in most research and development labs. Introduced by Robert Merrifield in 1963, it anchors the growing peptide chain to an insoluble resin bead. Each coupling and deprotection step is followed by a simple filtration wash, making the process highly amenable to automation. Modern synthesizers can run overnight cycles, building 50-residue sequences with minimal hands-on time.
LPPS builds peptides entirely in solution. It is better suited for short dipeptide or tripeptide fragments and is still preferred in certain GMP manufacturing contexts where solvent consumption and reagent cost per gram are tightly controlled. Fragment condensation, where larger protected segments are coupled together, is a hybrid strategy that borrows from both approaches.
| Feature | SPPS | LPPS |
|---|---|---|
| Automation potential | High | Low to moderate |
| Best for sequence length | Medium to long (5-50+ residues) | Short (2-10 residues) |
| Purification ease | Wash and filter between steps | Requires extraction/precipitation |
| Scale-up for GMP | Challenging but achievable | Often preferred for industrial scale |
| Side reaction control | Moderate, method-dependent | Generally easier for short fragments |
| Typical applications | R&D, discovery, custom synthesis | Fragment condensation, bulk APIs |
Hybrid approaches are gaining ground for complex therapeutic peptides. A common strategy is to synthesize protected fragments via SPPS, then condense them in solution to minimize epimerization risk at critical stereocenters. For teams tracking peptide research trends in 2026, hybrid synthesis is one of the faster-moving areas in process chemistry.
Pro Tip: In an R&D setting, default to Fmoc-SPPS for initial sequence screening and feasibility work. Switch to LPPS or hybrid fragment condensation only when you have identified your lead sequence and need to optimize cost-per-gram for scale-up. Changing strategy mid-discovery adds timeline risk without proportional benefit.
Critical steps and challenges in the synthesis process
Knowing the main methods, let’s dig deeper into the practical chemistry, the steps taken and the real-world obstacles encountered in the lab.
Key workflow steps in a standard Fmoc-SPPS run:
- Resin selection and loading (Wang, Rink amide, or Cl-Trt based on C-terminal requirement)
- Fmoc deprotection with piperidine in DMF
- Amino acid coupling using activating reagents (HATU, HBTU, or DIC/Oxyma)
- Capping unreacted amines with acetic anhydride to cap deletion sequences
- Iterative deprotection and coupling cycles until full sequence is assembled
- Global side-chain deprotection and resin cleavage with TFA cocktail
- Precipitation, crude peptide isolation, and RP-HPLC purification
- Analytical characterization by LC-MS and amino acid analysis
Two chemical protection strategies define the field. Fmoc (9-fluorenylmethyloxycarbonyl) protection uses mild base for alpha-amine deprotection and is compatible with acid-labile side-chain protecting groups. Boc-SPPS uses strong acid (HF) for both deprotection and cleavage, making it less common in standard labs but still critical for sequences prone to aspartimide formation or for synthesizing peptides containing acid-sensitive modifications.
Aspartimide formation is one of the most consequential side reactions in Fmoc-SPPS. It occurs when the Asp side-chain carboxyl attacks the backbone amide nitrogen during piperidine treatment, forming a cyclic imide that generates multiple truncated byproducts. Aspartimide prevention strategies include using backbone amide protection, adding 0.1 M HOBt to the piperidine solution, or switching to Asp(OtBu) with optimized deprotection conditions.

The waste problem is real. Typical peptide synthesis generates between 3,000 and 15,000 kg of chemical waste per kilogram of final peptide product. That number reflects the cumulative DMF washes, TFA cleavage volumes, and HPLC solvent consumption across a full synthesis and purification campaign.
Pro Tip: For sequences containing Asp-Gly or Asp-Ser motifs, add 0.1 M HOBt to every piperidine deprotection step from the start. Retrofitting this fix after you have already seen aspartimide peaks in your crude LC-MS adds days of troubleshooting. Proactive protection is always faster. For broader lab safety considerations, peptide safety tips are worth reviewing before scaling up TFA-heavy workflows.
Green chemistry and the future of peptide synthesis
With process challenges in mind, the conversation has shifted toward making peptide synthesis safer, greener, and smarter.
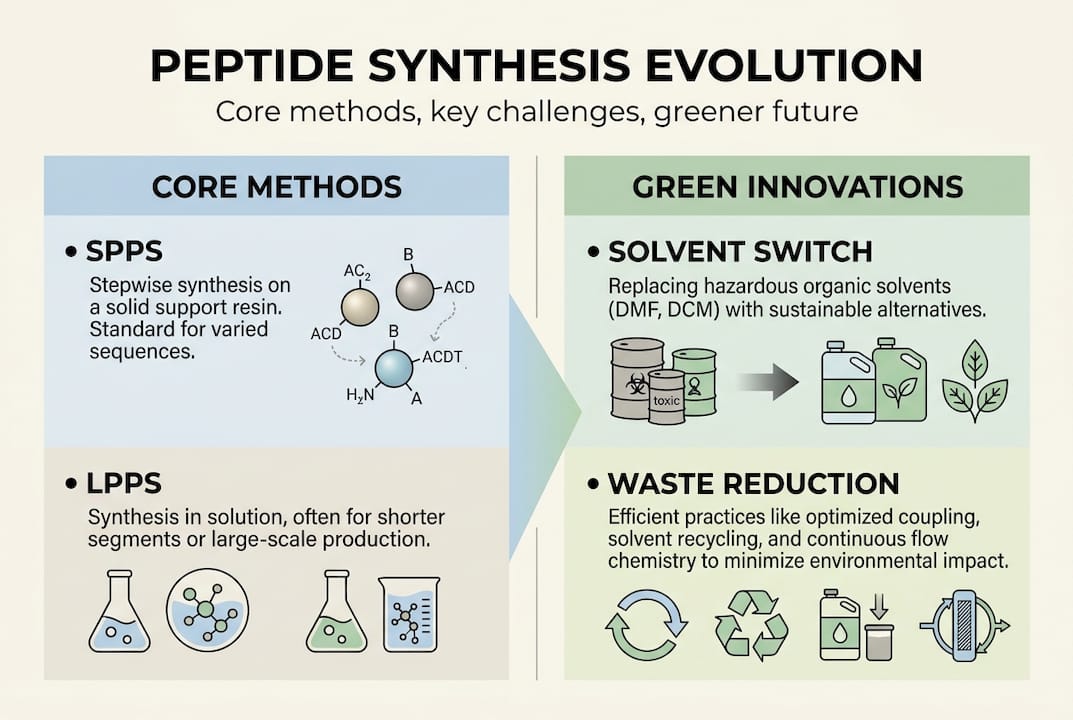
SPPS relies heavily on toxic solvents like DMF (dimethylformamide) and NMP (N-methyl-2-pyrrolidone), both of which are classified as substances of very high concern under REACH regulations. Regulatory pressure from the European Chemicals Agency and the FDA’s push for greener manufacturing are forcing process chemists to find alternatives now, not later.
| Green approach | Traditional baseline | Improvement |
|---|---|---|
| Cyrene as DMF replacement | DMF (toxic, regulated) | Lower toxicity, biodegradable |
| Aqueous SPPS | 100% organic solvent | Reduced solvent waste by up to 50% |
| Micellar catalysis | Organic coupling conditions | Water-compatible, lower waste |
| Microwave-assisted synthesis | Conventional heating | Faster cycles, higher yields |
| AI-guided sequence design | Trial-and-error optimization | Fewer synthesis failures upfront |
Emerging sustainable approaches include aqueous SPPS, micellar catalysis using surfactants like TPGS-750-M, and AI-guided peptide design that reduces the number of failed synthesis attempts before a viable sequence is identified. These are not theoretical proposals. Several are already in use at contract manufacturing organizations working on GLP-1 analog pipelines.
Key benefits of sustainable synthesis approaches:
- Reduced occupational exposure to classified hazardous solvents
- Improved regulatory compliance for GMP manufacturing audits
- Lower solvent procurement and disposal costs per batch
- Higher coupling efficiency in some aqueous-compatible systems
- Better alignment with ESG reporting requirements for biopharma companies
“The next decade will see AI-assisted peptide design and green solvent systems converge to make synthesis faster, cleaner, and more predictable than any previous generation of process chemists could have anticipated.”
For researchers tracking where the field is heading, research trends in peptide synthesis and peptide performance evidence both offer context on how these process innovations are translating into real-world therapeutic outcomes.
What most professionals miss about peptide synthesis
Here is the uncomfortable truth most training programs skip: method selection is almost always driven by institutional habit, not by rational analysis of the end application. Labs default to Fmoc-SPPS because that is what the last postdoc used. That inertia costs time and money.
The correct starting point is always the end use. A diagnostic imaging peptide needs isotopic purity above everything else. A GMP drug substance needs a defensible, scalable process. A screening library needs throughput. Each of those goals points toward a different synthesis strategy, and none of them automatically points to standard Fmoc-SPPS.
Automation is another area where efficiency can mask real problems. High-throughput synthesizers produce crude peptides fast, but purity profiles are often worse than manual synthesis for difficult sequences. Automation does not solve chemistry. It scales it, including the failures.
The professionals who consistently get the best outcomes treat method selection as a design decision, not a default. They also integrate green chemistry practices early, not as a compliance checkbox but because cleaner reactions genuinely produce better yields. For practical guidance on improving peptide outcomes, starting from the application and working backward is the framework that actually holds up.
Pro Tip: Before committing to a synthesis strategy, write out the three most important quality attributes for your final peptide. Then choose the method that optimizes for those attributes specifically, not the method that is easiest to set up.
Explore advanced peptide solutions and resources
Peptide synthesis sits at the intersection of chemistry, biology, and regulatory science, and staying current across all three is genuinely demanding work. The resources at Primegen Labs are built for exactly this audience.
Whether you are evaluating evidence-based peptide benefits for a new therapeutic program, working through the fundamentals with an in-depth peptide guide, or tracking the latest in peptide research to stay ahead of where the field is moving, Primegen Labs provides science-grounded content designed for researchers and professionals who need accuracy, not approximations. Explore the full resource library to support your next synthesis project or research program.
Frequently asked questions
What is peptide synthesis in simple terms?
Peptide synthesis is the laboratory process of chemically joining amino acids to create peptides, which serve as key building blocks for drugs and therapeutic agents.
What is the difference between SPPS and solution-phase synthesis?
SPPS attaches peptides to a solid resin for easy automation, while solution-phase synthesis builds peptides in a liquid; SPPS is typically used for longer or more complex sequences where iterative washing simplifies purification.
Why is peptide synthesis important for therapeutic development?
It allows precise manufacture of custom peptides that cannot be extracted from natural sources in usable quantities, enabling the creation of advanced drugs like GLP-1 agonists and cancer immunotherapy agents.
What are the biggest environmental challenges in peptide synthesis?
The process generates large amounts of chemical waste and uses hazardous solvents like DMF; greener methods including aqueous SPPS and micellar catalysis are now being developed to address these issues at scale.
