How peptides work: Mechanisms that drive fitness and weight loss

TL;DR:
- peptides are complex molecules with effects varying by receptor binding, tissue, dose, and modifications, not interchangeable ones.
- Understanding peptide mechanisms, receptor distribution, and chemical modifications enables precise protocol design and realistic client expectations.
Most fitness and nutrition professionals know peptides “do something” to appetite, muscle growth, or metabolism, but that generic framing is exactly where costly protocol mistakes begin. Peptides are not interchangeable molecules with uniform effects. Each one binds to specific receptors, triggers distinct intracellular cascades, and produces outcomes that vary by tissue, dose, and client physiology. Understanding these mechanisms at a functional level is what separates guesswork from precision, and it is the difference between a protocol that consistently delivers and one that underwhelms.
Table of Contents
- What peptides are and how they are made
- Core mechanisms: How peptides trigger responses in the body
- GLP-1 and weight loss peptides: A case study in multi-pathway action
- Chemical modifications: How design affects peptide action and outcomes
- A practitioner’s view: What most guides miss about peptides in weight loss and performance
- Next steps: Applying real peptide science to your practice
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Peptide actions are receptor-driven | Peptides work by binding to specific cell surface receptors that trigger cellular responses. |
| Central and peripheral pathways matter | A single peptide can impact multiple tissues and systems, not just one function or organ. |
| Modification boosts effectiveness | Chemical tweaks improve peptide stability, absorption, and real-world performance. |
| Practical protocols require mechanism matching | Selecting and recommending peptides for clients means understanding the underlying mechanism and desired outcome. |
What peptides are and how they are made
Before working with peptides in a client context, you need a clear picture of what you are actually dealing with at the molecular level. Peptides are short chains of amino acids, typically between 2 and 50 residues, cleaved from larger precursor proteins through a process called proteolytic processing. This is not a passive cutting step. The body precisely controls which fragments are released, when, and where.
What makes bioactive peptide functions so varied is the layer of post-translational modifications (PTMs) applied after the initial protein is synthesized. As research in Cell Trends confirms, peptides are produced via PTMs00063-5) that are critical for determining which bioactive forms exist and what functions they ultimately perform. In other words, the raw sequence is only part of the story.
Here is a breakdown of the most clinically relevant PTMs and what each one does:
| Modification | What it does | Practical impact |
|---|---|---|
| Amidation | Adds an amide group to C-terminus | Increases receptor binding affinity and stability |
| Sulfation | Adds sulfate to tyrosine residues | Critical for receptor recognition (e.g., cholecystokinin) |
| Pyroglutamylation | Cyclizes N-terminal glutamine | Protects from enzymatic degradation |
| Phosphorylation | Adds phosphate group to serine/threonine | Regulates activity and signaling interactions |
| Acylation | Attaches fatty acid chains | Alters membrane interaction and half-life |
| Disulfide bonds | Bridges cysteine residues | Locks conformation for structural stability |
Key takeaways for practitioners:
- Stability matters. A peptide without protective modifications is rapidly degraded in plasma, often within minutes.
- Conformation drives selectivity. The 3D shape of a peptide determines which receptor it binds. Minor structural changes can completely alter target specificity.
- PTMs are not cosmetic. They are functionally essential, not decorative add-ons made in manufacturing.
Understanding this foundation helps you evaluate which peptide products are likely to behave as claimed versus those that are poorly designed or inadequately stabilized for practical use.
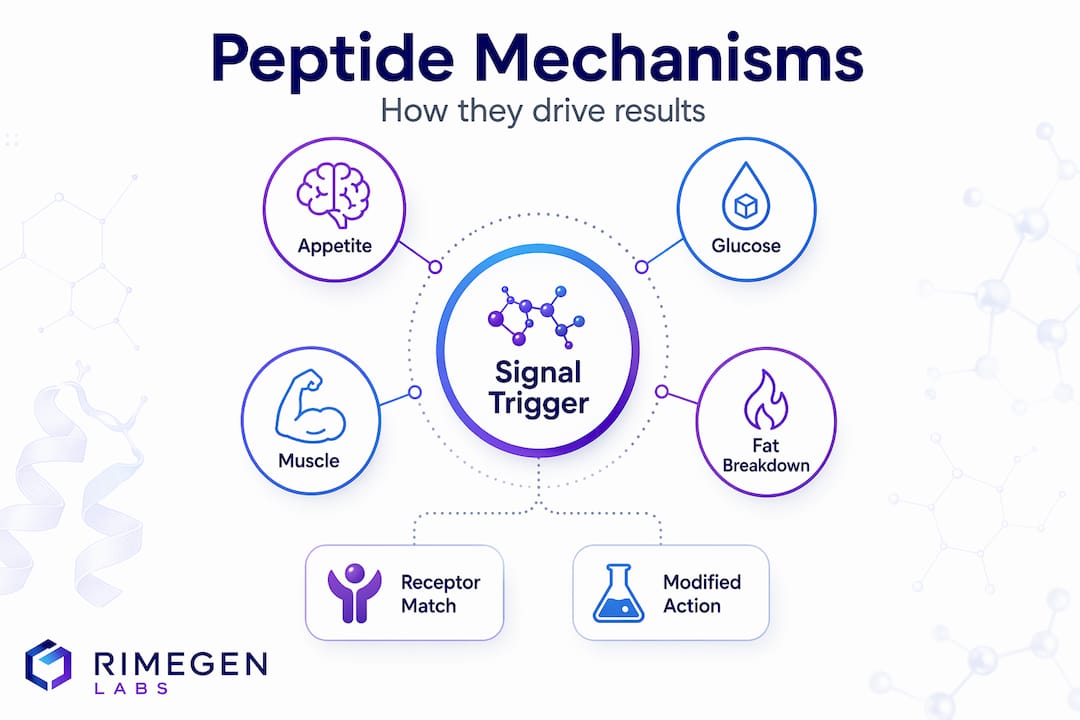
Core mechanisms: How peptides trigger responses in the body
Structure tells you what a peptide is. Mechanism tells you what it does and why. Most peptide actions begin the moment the molecule docks with a receptor on the surface of a target cell. As peptide hormone signal transduction research confirms, receptor binding triggers intracellular signaling cascades through a few major receptor classes, each with distinct downstream consequences.
The three main receptor types relevant to peptide action are:
- G protein-coupled receptors (GPCRs): The largest and most diverse receptor family. When a peptide binds, the GPCR activates a G protein, which then regulates second messengers like cyclic AMP (cAMP) or inositol triphosphate (IP3). This cascade amplifies the signal far beyond what a single binding event would suggest.
- Enzyme-coupled receptors: These include receptor tyrosine kinases, which become enzymatically active upon peptide binding and directly phosphorylate downstream targets. Insulin receptor signaling operates this way, making this class particularly relevant for metabolic protocols.
- Ion-channel-coupled receptors: Peptide binding directly opens or closes ion channels, producing rapid electrical or concentration-based cellular changes. These are especially important in neural signaling contexts.
| Receptor class | Signal mechanism | Example peptide | Clinical relevance |
|---|---|---|---|
| GPCR | cAMP / IP3 second messengers | Secretin, GLP-1 | Metabolic regulation, satiety |
| Enzyme-coupled | Tyrosine kinase phosphorylation | Insulin, IGF-1 | Glucose uptake, muscle anabolism |
| Ion-channel-coupled | Ionic flux changes | Neuropeptides | Pain, appetite, neural signaling |
One critically underappreciated point: the same peptide can yield fundamentally different effects depending on which tissues it reaches. Secretin, for instance, binds GPCRs in pancreatic ductal cells and triggers cAMP production, stimulating bicarbonate secretion. In a different tissue, that same signaling molecule plays a different role entirely. For appetite pathway peptides, this tissue-specificity is the entire mechanistic story behind why dosing route and timing matter so much in practice.
“Receptor distribution is the map. Without knowing where a receptor sits, you cannot predict what a peptide will do in a given tissue. This is not theory. It is the core variable in every protocol decision.”
Pro Tip: When reviewing peptide research, always check whether receptor expression data is available for the target tissue. A peptide with a great hypothalamic signal may do very little in peripheral muscle if the receptor is not expressed there at meaningful levels.
GLP-1 and weight loss peptides: A case study in multi-pathway action
GLP-1, or glucagon-like peptide-1, is arguably the most instructive example available right now for understanding how weight-loss peptides work through layered, system-wide mechanisms rather than a single pathway.
GLP-1 receptor agonists act in multiple anatomical compartments simultaneously. According to research in the American Journal of Medicine, GLP-1 RA mechanisms00059-2/fulltext) include enhanced glucose-dependent insulin secretion, inhibition of glucagon release, slowing of gastric emptying, and suppression of appetite via hypothalamic circuitry. That is four distinct mechanisms in one molecule.
From a practitioner standpoint, here is how those mechanisms map to client outcomes:
- Glucose-dependent insulin secretion: Prevents hyperglycemia post-meal without significant hypoglycemia risk, making these peptides safer in metabolically compromised clients than older drug classes.
- Glucagon suppression: Reduces hepatic glucose output, which lowers fasting blood glucose and supports caloric deficit sustainment.
- Gastric emptying delay: Slows nutrient absorption, extends satiety duration, and flattens postprandial glucose spikes.
- Hypothalamic appetite suppression: Reduces caloric drive centrally, which is the mechanism most clients and practitioners focus on, but far from the only one.
Statistic worth knowing: Clinical trials on GLP-1 receptor agonists have reported average weight reductions of 12 to 22 percent of body weight over 68 weeks in adults with obesity, outcomes that exceed lifestyle intervention alone by a significant margin. This is not appetite suppression doing all the work. It is the combination of all four mechanisms operating concurrently.
As Nature Reviews Drug Discovery confirms, GLP-1-based therapies engage multiple mechanisms beyond appetite reduction, including pharmacokinetic factors that shape how and when these effects are expressed across the dosing cycle.
For weight loss peptide strategies to work at their highest potential, practitioners need to understand which mechanisms are most relevant to a specific client’s primary barrier. Is the issue uncontrolled appetite, poor glucose regulation, or rapid gastric transit? Each answer suggests a different prioritization in the protocol design.
Understanding peptides and fitness performance through the GLP-1 model also helps practitioners anticipate potential side effects, since mechanisms like gastric slowing directly explain the nausea reported early in treatment.
| Client profile | Primary relevant mechanism | Protocol focus |
|---|---|---|
| High-appetite, normal glucose | Hypothalamic suppression | Timing around high-risk eating windows |
| Post-meal glucose spikes | Insulin potentiation + glucagon inhibition | Dosing relative to meal schedule |
| Slow satiety onset | Gastric emptying delay | Meal composition and portion guidance |
| Combined metabolic dysfunction | All four simultaneously | Comprehensive protocol with monitoring |
Chemical modifications: How design affects peptide action and outcomes
Natural peptides evolved for speed and turnover. The body produces them, they signal, and they are cleared quickly. That is ideal for biological homeostasis but problematic for therapeutic use, where you need predictable, sustained action across a dosing window.
Most naturally occurring peptides have plasma half-lives measured in minutes. Without modification, a peptide administered subcutaneously might be degraded before it reaches its target receptor in meaningful concentrations. This is why peptide optimization strategies are not optional engineering exercises. They are the foundation of a peptide that actually works clinically.
According to a Springer review on peptide modification strategies, the key pharmacology constraints addressed through chemical modification include stability, half-life, solubility, pharmacokinetic behavior, and delivery optimization. The modifications that matter most in practice include:
- PEGylation: Attaching polyethylene glycol chains to increase molecular size, which reduces renal clearance and extends half-life dramatically.
- Lipidation (fatty acid conjugation): Allows peptides to bind albumin in circulation, acting as a depot that slowly releases the active molecule. Semaglutide uses this approach to achieve its once-weekly dosing window.
- D-amino acid substitution: Replacing natural L-amino acids with their mirror D-form makes the peptide resistant to proteolytic enzymes that would normally break it down.
- Cyclization: Creating a ring structure, either through disulfide bonds or other linkers, which improves both stability and receptor binding specificity.
- Backbone modification: N-methylation and peptoid substitutions alter the peptide backbone in ways that resist enzymatic attack without significantly impairing receptor interaction.
Pro Tip: When evaluating a peptide supplement or therapeutic product for client recommendation, look specifically for disclosed modifications. Products that offer no modification data or claim to deliver unmodified peptides orally should be scrutinized carefully. Unmodified peptides rarely survive the gastrointestinal environment intact.
Understanding peptide bioavailability for fitness is especially critical when helping clients navigate supplement choices versus prescription options. The modification tier of a peptide directly determines its bioavailability ceiling.

A practitioner’s view: What most guides miss about peptides in weight loss and performance
Most peptide guides stop at mechanism and treat it as the destination. In practice, mechanism is just the starting point. Here is what deserves more direct attention in professional conversations about peptides.
First, multi-compartment signaling is the rule, not the exception. As confirmed in Nature Reviews Drug Discovery, GLP-1 receptor signaling operates in both gut-peripheral and central nervous system compartments simultaneously. This means attributing a client’s outcome to a single pathway is usually an oversimplification. Weight change happens because of a cascade of concurrent signals, and adjusting the protocol requires thinking about all of them at once.
Second, evidence quality is wildly uneven across peptides. Some compounds have robust phase 3 human trial data and regulatory approval. Others have compelling rodent data and enthusiastic anecdotal reports. That gap matters enormously when you are advising a real person making real health decisions. Knowing current peptide regulations in 2026 is not optional. Regulatory status reflects both safety evidence and legal professional liability.
Third, pharmacokinetics determine whether mechanism ever becomes outcome. You can have a perfectly targeted peptide with flawless receptor specificity. If it is dosed incorrectly, delivered via a route that destroys it, or timed without reference to the client’s metabolic rhythms, you will see minimal effect. Understanding peptide supplement risks includes understanding that poor pharmacokinetic design is one of the most common sources of client disappointment, not mechanism failure.
Finally, compliance is mechanistic too. A peptide that requires daily injections and causes persistent nausea in the first four weeks will produce worse real-world outcomes than a mechanistically “inferior” peptide that clients actually take consistently. Match the dosing burden and side effect profile to the client’s tolerance and lifestyle before committing to a protocol.
Next steps: Applying real peptide science to your practice
For health and fitness professionals who want to move from theory to practice, access to curated, up-to-date resources is essential.
Applying peptide science well means more than reading mechanism papers. It means having reliable, evidence-based tools to evaluate products, design protocols, and educate clients accurately. At Primegen Labs, we build our resources specifically for practitioners who need that layer of depth, not oversimplified marketing summaries. Start with our science-backed peptide supplement guide to understand which compounds have the strongest evidence base, then move into our muscle growth peptide guide for performance-focused protocol design. The science is specific. Your resources should be too.
Frequently asked questions
How do peptides interact differently with muscle versus fat cells?
Peptide effects depend entirely on which receptors are expressed in a given tissue. As peptide signal transduction research confirms, certain peptides activate anabolic pathways in muscle while others primarily modulate lipolysis or fat storage in adipose tissue, based on receptor presence and downstream signaling machinery.
Are all weight-loss peptides acting through appetite suppression?
No. Peptides like GLP-1 work through at least four mechanisms simultaneously. GLP-1 receptor agonists00059-2/fulltext) engage glucose-dependent insulin secretion, glucagon suppression, gastric emptying delay, and central appetite pathways, making appetite suppression just one part of a much broader mechanistic picture.
What are the main challenges in peptide drug development?
The biggest hurdles are short half-life, rapid enzymatic degradation, and getting adequate concentrations to target tissues. Therapeutic peptide modifications address these constraints through strategies like PEGylation, lipidation, D-amino acid substitution, and cyclization.
Why do some supplements with peptides not show expected results?
If a peptide lacks protective chemical modifications, it is likely degraded in the gastrointestinal tract or bloodstream before reaching target receptors. Inadequate peptide stability and poor cellular uptake are among the most common formulation failures behind disappointing supplement outcomes.
